Siemens process control system first product with IEC 62443 security certification | Press | Company | Siemens
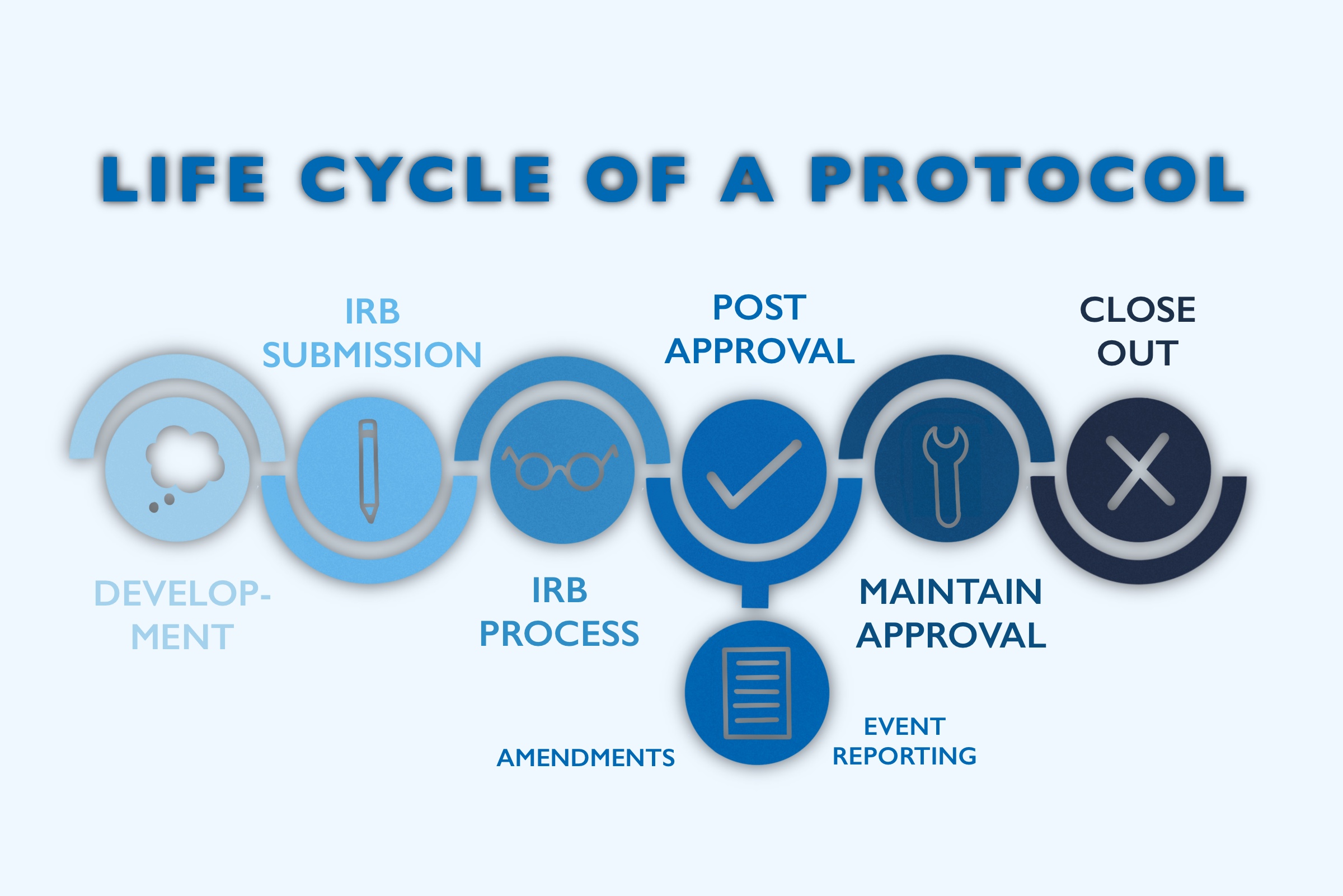
How do Institutional Review Boards (IRB) and Ethics Committees (EC) impact clinical trials? - Clincierge

Regulatory oversight of cell therapy in China: Government's efforts in patient access and therapeutic innovation - ScienceDirect
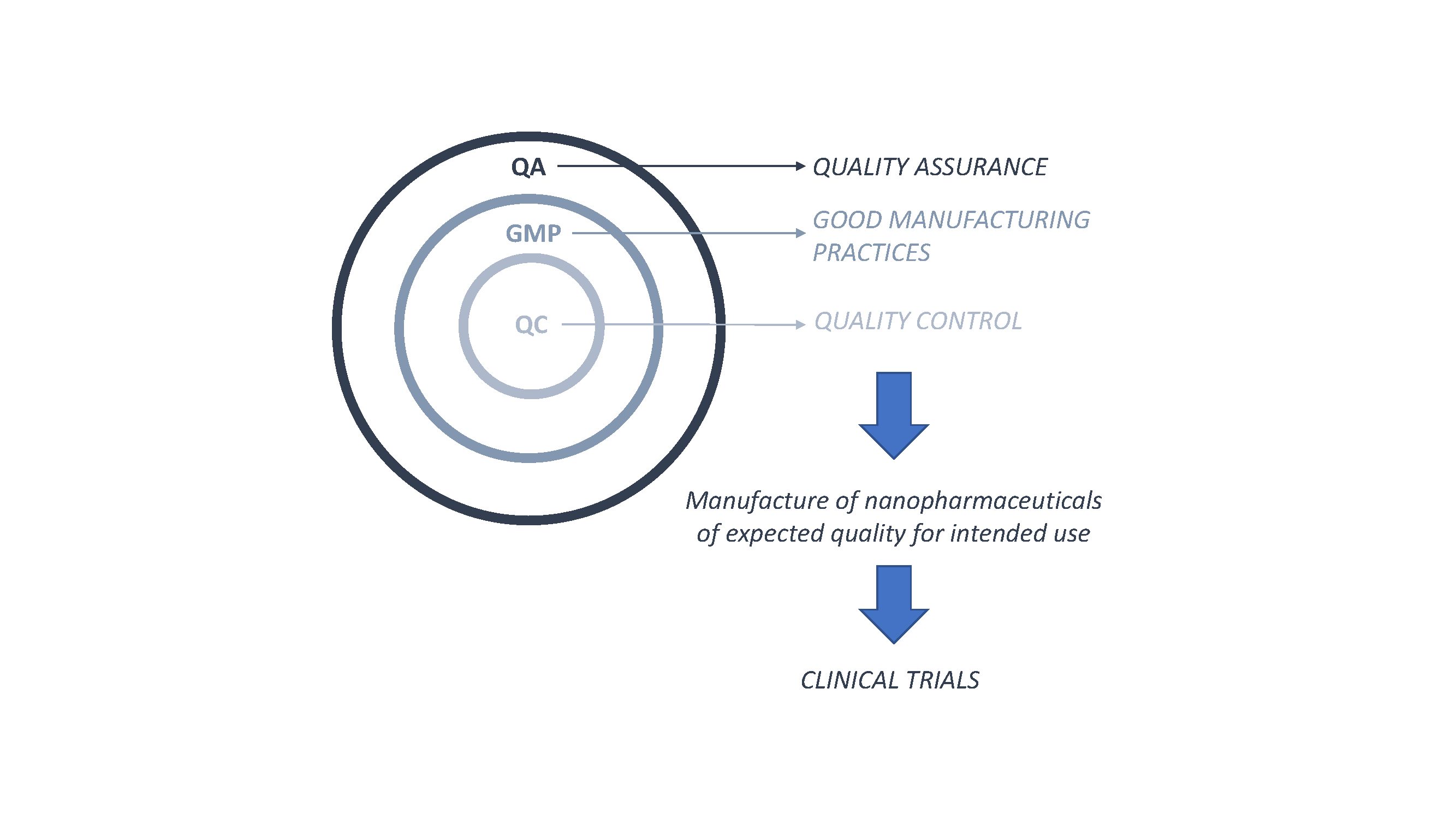
Pharmaceutics | Free Full-Text | Nanopharmaceutics: Part I—Clinical Trials Legislation and Good Manufacturing Practices (GMP) of Nanotherapeutics in the EU | HTML

PDF) Blended Discrete Trial Clinical Method to Enhance Performance in e-Learning Mathematics Courses | Omar Majid - Academia.edu